Vaccine manufacturer for the SARS-CoV-2 novel coronavirus, Pfizer, says it will advance its mRNA vaccine business beyond the virus which causes the disease the Chinese Communist Party-friendly World Health Organization dubbed Coronavirus Disease 2019 (COVID-19).
In a March 23 article by the Wall Street Journal, Pfizer CEO Albert Bourla said his company gained years of experience in the single year they worked on their COVID-19 vaccine, and are eager to expand their newly developed business model and manufacturing capability for mRNA vaccines beyond the SARS-CoV-2 virus.
Pfizer anticipates producing the Covid-19 shots for at least several years on the expectation that booster shots will be needed annually or every few years to maintain protection.
Pfizer CEO Albert Bourla on his company’s mRNA vaccine business
The WSJ article says Bourla declined to comment on which viruses Pfizer would target, or its plans for future vaccine sales, but did say “He hopes that mRNA can also be effective in areas where existing vaccines have uncomfortable side effects or provide subpar protection.”
Success
You are now signed up for our newsletter
Success
Check your email to complete sign up
The article says Pfizer first began experimenting with mRNA vaccines in 2018 when it partnered with BioNTech to make a flu vaccine based on BioNTech’s platform, which ultimately led to the two’s collaboration in the creation of the COVID-19 vaccine.
Bourla said, “We like working with BioNTech, but we don’t need to work with BioNTech. We have our own expertise developed.”
The article adds that Pfizer has secured “specialized raw materials for manufacturing” and “built a bespoke production network” because “making mRNA vaccines requires special equipment and raw materials that weren’t widely available prior to the pandemic. Pfizer scientists learned how to produce key ingredients for the shot that they once relied on suppliers to obtain.”
The Fact Sheet for the Pfizer-BioNTech vaccine says the ingredients are primarily mRNA instructions, a number of lipid nanoparticles, chemicals such as potassium chloride, and sucrose.
According to the article, Bourla also said “Pfizer anticipates producing the Covid-19 shots for at least several years on the expectation that booster shots will be needed annually or every few years to maintain protection.”
The new mRNA vaccines were developed on a highly accelerated timeline and differ from the traditional inactivated virus vaccines. While an inactivated virus vaccine attempts to generate antibodies by introducing a weakened or inert form of the virus into the human body, mRNA provides an instruction to your cells which causes ribosomes to produce only the spike protein from the SARS-Cov-2 virus, which it relies on to connect with and enter human cells in order to infect a host and survive against the human immune system.
By comparison, the human immune system’s natural generation of antibodies from exposure, and the portion of herd immunity which comes from it, would generate an antibody in response to the whole virus and all of its molecular chains, rather than just the spike protein targeted with the mRNA vaccine.
As seen on Pfizer’s website for this vaccine, the company states, “Unlike a traditional vaccine that uses inactivated, dead, or portions of actual virus to spur an immune response, mRNA delivers a message to your body’s cells via a lipid nanoparticle envelope that instructs the cells to generate the spike protein found on the surface of a coronavirus that initiates infection. Instructing cells to generate the spike protein spurs an immune response, including generation of antibodies specific to the SARS-CoV-2 spike protein.”
Moderna’s mRNA vaccine business ‘an operating system’
Pfizer’s competitor, Moderna, who uses the same mRNA concept but of an independently developed sequence and delivery mechanism, refers to their company’s mRNA as “an operating system” on their website.
“Recognizing the broad potential of mRNA science, we set out to create an mRNA technology platform that functions very much like an operating system on a computer. It is designed so that it can plug and play interchangeably with different programs. In our case, the ‘program’ or ‘app’ is our mRNA drug – the unique mRNA sequence that codes for a protein.”
Moderna continues, “Generally, the only thing that changes from one potential mRNA medicine to another is the coding region – the actual genetic code that instructs ribosomes to make protein.”
“Utilizing these instruction sets gives our investigational mRNA medicines a software-like quality. We also have the ability to combine different mRNA sequences encoding for different proteins in a single mRNA investigational medicine.”
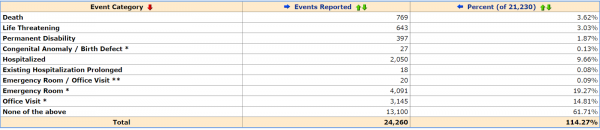
The U.S. Centers for Disease Control VAERS adverse reaction database has recorded a little over 21,000 reports for the Pfizer-BioNTech variant of the vaccine, including 4,091 emergency room visits, 2,050 hospitalizations, 769 deaths, 397 permanent disabilities, and 27 “congenital anomaly / birth defects as of March 24.
According to the National Vaccine Information Center, fewer than 1% of vaccine adverse events are reported. Still, the CDC disclaims on the VAERS search “Submitting a report to VAERS does not mean that healthcare personnel or the vaccine caused or contributed to the adverse event (possible side effect).”
Follow us on Twitter or subscribe to our email list







