Beijing has been promoting its “vaccine diplomacy” since last year, aiming to expand its diplomatic influences by providing easily accessible made-in-China vaccines. However, the efficacy of Chinese variants of the COVID-19 vaccine has now been widely questioned, especially against new mutations of the virus. Some countries that have high vaccination rates but are using Chinese vaccines are still experiencing serious outbreaks, showing the country’s offerings are not as good as China’s official propaganda.
The New York Times reported on June 22 that countries using the Chinese Communist Party’s (CCP) vaccines were not getting rid of the virus, but instead were struggling with a surge in positive cases.
The report cited data from the data tracking project Our World in Data, which showed that Seychelles, Chile, Bahrain, and Mongolia, where 50 to 68 percent of the population has been fully vaccinated in addition to a coverage rate higher than that of the United States, are among the top 10 countries with the most severe outbreaks of COVID-19. These four countries are all using vaccines made in China that come from either Sinopharm or Sinovac Biotech.
The University of Oxford’s previous statistics also shows that as of June 9, of the 10 countries with the highest number of confirmed cases per million people on a single day, nine countries have received a large number or part of the vaccine produced in China. In Seychelles, which has the highest infection rate, nearly 60 percent of the injections administered are Sinopharm; in Uruguay, 80 percent of the vaccines are Sinovac Biotech.
Mongolia has received millions of doses of Sinopharm vaccines through the CCP’s vaccine diplomacy initiatives. Now, despite 52 percent of the Mongolian population having been fully inoculated, on June 20, Mongolia saw 2,400 new cases of infection, four times the number of cases a month ago.
Success
You are now signed up for our newsletter
Success
Check your email to complete sign up
Bahrain was also one of the first countries to accept Chinese vaccines. The island country in the Persian Gulf approved the offerings for use before data from late-stage clinical trials were available, and has continued to report large numbers of positive cases in vaccine recipients.
In Seychelles, which relies heavily on the Sinopharm vaccine, the number of newly confirmed Covid-19 cases per million people reached 716 in mid-June. By comparison, in Israel, where the Pfizer vaccine is administered, the cases per million are down to under 5.
Meanwhile, a new variant of the virus is spreading in Indonesia. The Indonesian Medical Association’s Risk Mitigation Group says more than 350 doctors and health care workers recently began suffering from COVID-19, even though they were fully vaccinated with Sinovac. Between February and June 7, 61 physicians died from COVID-19, in which 10 of them were given the Chinese-made vaccine, the Association said.
Kenneth Mak, director of medical services in Singapore, said at a press conference that evidence from other countries showed people who had taken the Sinovac vaccine were facing “A significant risk of vaccine breakthrough.”
Singapore has been offering Sinovac for the first time to the public since June 18. Demand has been high among Chinese nationals in the country who fear only Chinese vaccines will be accepted as valid if they return to the mainland.
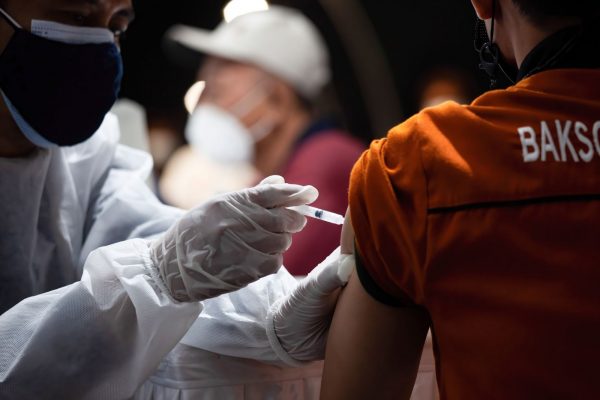
The New York Times noted that vaccine disparities may have led to the division of countries into three groups in the post-pandemic era: the first group of wealthy countries that used Pfizer-BioNTech and Modena vaccines, the second group of poorer countries that did not have enough vaccine to mass vaccinate their citizens, and “China, as well as the more than 90 nations that have received the Chinese shots, may end up in the third group, contending with rolling lockdowns, testing, and limits on the day.
“Economies could remain held back. And as more citizens question the efficacy of Chinese doses, persuading unvaccinated people to line up for shots may also become more difficult,” posits the author.
Despite controversies about the efficacy of China-made vaccines, Beijing’s Foreign Ministry claimed in a statement that the country’s vaccine “is aimed at stopping serious illnesses rather than the spread of the virus, and is recognized for its safety and efficacy.” The statement continues that China has not found a connection between the vaccine and the recent outbreak while relying on WHO data that says vaccination rates in those countries are not yet high enough to prevent an outbreak and that further preventive measures must be maintained.
Little crucial clinical data has been made public about the Chinese vaccines. Neither Sinopharm nor Sinovac responded to inquiries from the New York Times and other Chinese media regarding the controversy.
Nikolai Petrovsky, a professor at the College of Medicine and Public Health at Flinders University in Australia, told NYT that all evidence suggests that the Sinopharm vaccine has done little to contain the spread of the disease. He also mentioned that the Chinese vaccines may have allowed the infected person to develop few or no symptoms and still transmit the virus to others.
“I think that this complexity has been lost on most decision makers around the world,” he said.
William Schaffner, Medical Director of the National Foundation for Infectious Diseases at Vanderbilt University makes a similar point to NYT, “The efficacy rates of Chinese shots could be low enough to sustain some transmission, as well as create illness of a substantial amount in the highly vaccinated population, even though it keeps people largely out of the hospital,” he said.
The recent pandemic situation in France has shown signs of slowing down. Recently, restrictions on the entry of vaccinated foreign visitors have relaxed. Those who have received the vaccine can now enter France without providing an “urgent reason.” However, only Modena, Pfizer BNT, AstraZeneca, and J&J vaccines are recognized by the French side.
The Chinese media quoted the Chinese Embassy in France on June 15 as stressing that the Chinese vaccine had been included in the WHO’s emergency use list last month and should be recognized by the French government. The CCP claimed that “reciprocal sanctions” would be applied against France, and that Beijing would not recognize the non-Chinese vaccines received by French people entering China in retaliation.
However, the Chinese Sinovac and Sinopharm vaccines have not yet been approved for use by the European Medicines Agency (EMA).















