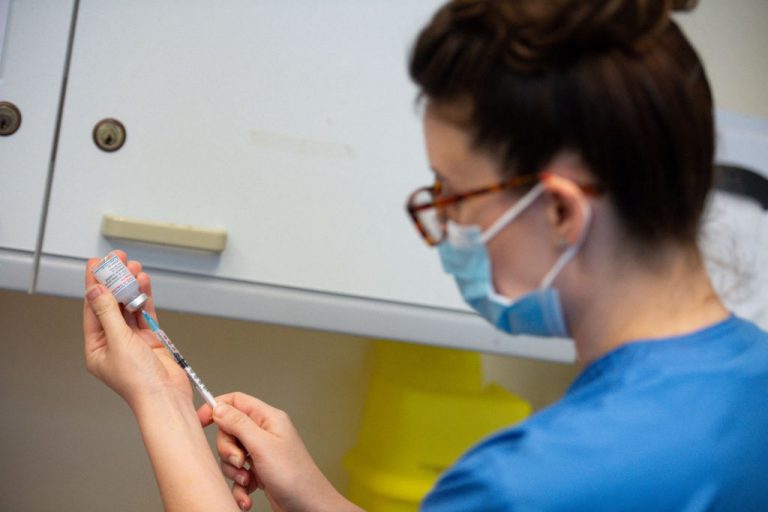
On June 30, the UK Department of Health and Social Care announced that millions of people “most vulnerable” to Coronavirus Disease 2019 (COVID-19) would be provided with a booster shot in September this year. The department sought to stave off new coronavirus variants and prepare for the upcoming winter season.
The announcement follows interim advice by the Joint Committee on Vaccination and Immunisation (JCVI), which argued for booster vaccines starting in September. The booster COVID-19 vaccination campaign will coincide with the annual flu vaccination campaign. The final JCVI advice on the issue will be published before September, and the third booster shot will be distributed in two stages.
In the first stage, the following people will be offered the third COVID-19 shot and influenza vaccine: (a) those aged 16 and older who are immunosuppressed or clinically considered extremely vulnerable, (b) adults aged 70 and older, (c) people who live in old age residential homes, and (d) frontline healthcare and social care workers.
People eligible in the second stage include (a) adults aged 50 and older, (b) adults between the age of 16 and 49 who are in influenza or COVID-19 at-risk group, (c) adults living together with immunosuppressed people.
“We welcome this interim advice, which will help us ensure we are ready in our preparations for autumn. We look forward to receiving the committee’s final advice in due course. We need to learn to live with this virus,” Sajid Javid, Health and Social Care Secretary, said in a statement.
Success
You are now signed up for our newsletter
Success
Check your email to complete sign up
The idea of a third booster shot is being pushed aggressively by COVID-19 vaccine manufacturers. In an interview with CNBC in April, Pfizer CEO Albert Bourla stated that people would “likely” need to receive a third booster shot 12 months after being fully vaccinated and that an annual jab might be needed.
However, the vaccination efforts have met with strong criticism. “There is zero, and I mean zero, evidence to suggest that that is the case… It’s completely inappropriate to say that we’re likely to need an annual booster, because we have no idea what the likelihood of that is,” Dr. Tom Frieden, former director of the U.S. Centers for Disease Control and Prevention (CDC), said to Reuters.
Some scientists have raised concerns that pharma executives rather than health specialists are influencing people’s thoughts on COVID-19 boosters. Kate O’Brien, director of the Department of Immunization, Vaccines, and Biologicals at the World Health Organization (WHO), stated that they have not seen any data supporting the need for booster doses.
According to the UK’s Yellow Card system, 298,081 adverse events from COVID-19 vaccines have been reported as of June 23rd. The AstraZeneca vaccine was associated with 211,339 reports, the Pfizer-BioNTech vaccine with 79,296 reports, and the Moderna vaccine with 6,583 reports.
The number of daily new confirmed COVID-19 cases per million people in the UK was at 395.71 on July 2, up from between 35 and 55 during the April to May period. Between April 1 and July 2, the percentage of fully vaccinated people in the UK jumped from 7.3 percent to 49.2 percent.
Vaccinating children
In April, The Sun reported that the UK government was considering giving COVID-19 jabs to kids as young as 12 years of age. In an open letter published in May, a group of more than 40 doctors, scientists, and medics criticized the move.
The letter pointed out that children “are at almost no risk from COVID-19,” with an extremely low death rate of 1 in 2.5 million. In addition, children do not transmit the virus “as readily as adults.”
The letter raised concerns about VAERS reporting in the United States, which showed that there have been a number of deaths among children who were given a COVID-19 vaccine. The experts warned that the neurological and immunological systems of children are still developing, which makes them “more vulnerable” to vaccine adverse events.
“We conclude that it is irresponsible, unethical and indeed, unnecessary, to include children under 18 years in the national COVID-19 vaccine rollout. Clinical trials in children also pose huge ethical dilemmas, in light of the lack of potential benefit to trial participants and the unknown risks. The end of the current Phase 3 trials should be awaited as well as several years of safety data in adults, to rule out, or quantify, all potential adverse effects,” the letter said.
A BBC report published on June 16 stated that the government’s decision to vaccinate all 12 to 17-year-old children is unlikely to be backed by the country’s vaccine experts anytime soon. However, certain groups of kids might still be vaccinated.