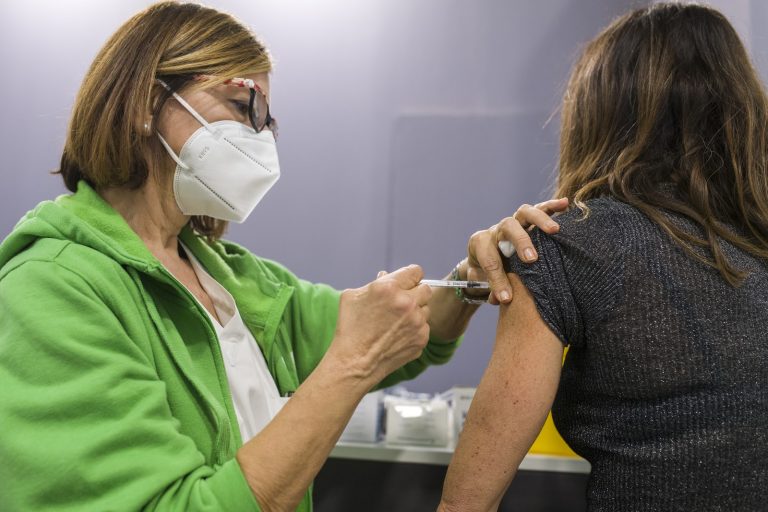
A Pfizer executive has said the company might raise its coronavirus vaccines’ price given the “significant opportunity.” The suggestion was made by Frank A D’Amelio, CFO & executive VP of global supply for Pfizer, at a virtual international healthcare conference hosted by Barclays.
D’Amelio’s prediction is based on the coronavirus becoming endemic. That means that the virus will always remain globally, and annual shots may be required to protect people from infection. The vaccine could be a cash cow for Pfizer.
“If you look at how current demand and current pricing is being driven, it’s not being driven by what I’ll call normal market conditions, normal market forces. It’s been driven by kind of the pandemic state that we’ve been in and the needs of governments to secure doses from the various vaccine suppliers,” D’Amelio said at the conference. Once it becomes endemic, regular market forces and conditions will kick in. Then factors like booster ability, efficacy, and clinical utility become essential.
Vaccine price hike
Last month, John LaMattina, a reporter at Forbes, warned that COVID-19 vaccines could be priced higher in the future. Presently, the most expensive two-dose Pfizer coronavirus vaccine costs $39, the equivalent of a quadrivalent flu shot.

Success
You are now signed up for our newsletter
Success
Check your email to complete sign up
However, the price of COVID-19 vaccines could soon triple or quadruple up to $156. Such a price hike could hurt the company’s public image; Pfizer executives could even be called to testify about the increase at the Capitol.
Pfizer’s contract with the U.S. government requires it to provide 100 million doses by the end of March. But the company is on track to deliver 120 million shots by that time.
By May, the target is to deliver 200 million vaccines. For the full year, the original goal was to deliver 1.3 billion doses. However, D’Amelio said they are likely to produce and distribute two billion doses and maybe even more. The company is getting more orders from the international market.
Brazil has ordered 100 million doses of the vaccine. Japan will be receiving 100 million shots by June. The South African drug regulator SAHPRA was approved to use the vaccine for emergency use. There have been talks about the company producing vaccines in India to boost supply capability.
However, a Pfizer spokesperson dismissed such reports. It keeps its production facilities in the United States and Europe for now. But once the pandemic supply phase is over, the company will “evaluate all additional opportunities available.”
Selling Chinese business
Pfizer recently announced that it is stopping biosimilar products in communist China. Biosimilars, or biologics, are officially approved versions of an original medical product manufactured after the expiration of the original product’s patent.
Biologic drugs are made from living organisms and are more expensive and tougher to make than conventional medications formulated from chemicals. Pfizer will be selling a business unit in Hangzhou to WuXi Biologics Inc.
“The site was planned to manufacture three biosimilars for the China market… Pfizer commercially and technically evaluated other products for the site, but none reached the level of activity for the scale of the site,” the company told Reuters. The sale is expected to be concluded in the first half of this year. The value of the deal has not been revealed.
Follow us on Twitter or subscribe to our weekly email