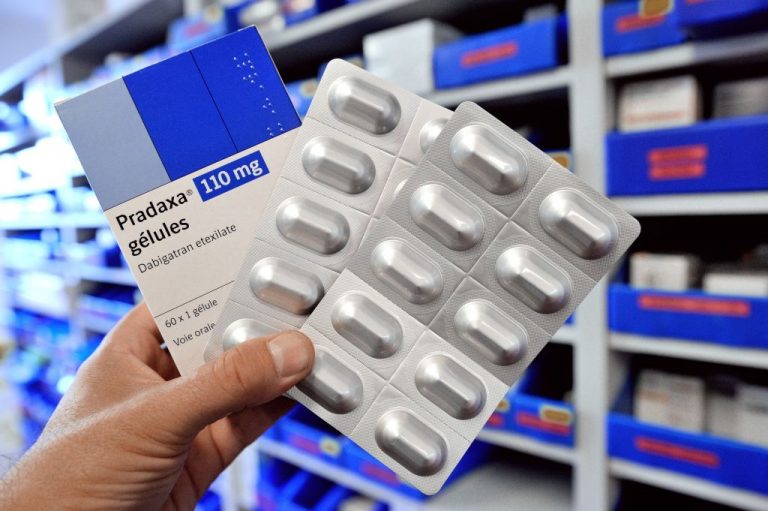
The U.S. Food and Drug Administration quietly approved an orally-administered blood thinner for children to combat venous thromboembolism just months before the rollout of Coronavirus Disease 2019 (COVID-19) novel gene therapy vaccines in kids aged 5 to 11 are set to start.
The FDA approved Pradaxa oral pellets, described as a drug used “to treat children 3 months to less than 12 years old with venous thromboembolism (a condition where blood clots form in the veins) directly after they have been treated with a blood thinner given by injection for at least five days,” according to a June 21 press release.
“The FDA also approved Pradaxa oral pellets to prevent recurrent clots among patients 3 months to less than 12 years old who completed treatment for their first venous thromboembolism.”
The release continues, “Pradaxa is the first FDA-approved blood thinning medication that children can take by mouth; the only other approved blood thinning medication for children is given by injection.”
The FDA says Pradaxa was first approved in 2010 “to reduce the risk of stroke and systemic embolism in adult patients with non-valvular atrial fibrillation.”
Clotted blood
Success
You are now signed up for our newsletter
Success
Check your email to complete sign up
In a CDC web page titled What is Venous Thromboembolism?, the agency describes the phenomenon in the following way: “Deep Vein Thrombosis and Pulmonary Embolism (DVT/PE) are often underdiagnosed and serious, but preventable medical conditions…Deep vein thrombosis (DVT) is a medical condition that occurs when a blood clot forms in a deep vein. These clots usually develop in the lower leg, thigh, or pelvis, but they can also occur in the arm.”
READ MORE:
- CDC Update on Neurological, Blood, Heart Inflammation Adverse Events From COVID-19 Vaccines
- Twitter Labels Obituary of 37-Year-Old Seattle Mother Who Died From COVID Vaccine Blood Clots ‘Misleading’
- Merck’s COVID Pill ‘Will be Incorporated in the DNA’: Researcher
- Trio of Pfizer Scientists Expose Surveillance and Profit-driven Corporate Culture, Virtues of Natural Immunity on Hidden Camera
The most significant complication arising from DVT is noted as occurring when “a part of the clot breaks off and travels through the bloodstream to the lungs, causing a blockage called pulmonary embolism (PE).”
“If the clot is small, and with appropriate treatment, people can recover from PE. However, there could be some damage to the lungs. If the clot is large, it can stop blood from reaching the lungs and is fatal.”
The CDC states that the most significant risk factors contributing to DVT are injury to a vein, slow blood flow, increased estrogen, chronic illnesses such as Chron’s, cancer, lung disease, or heart disease, as well as general risk factors such as obesity, age, catheters located in central veins, and clotting disorders.
The FDA release warns that Pradaxa may include side effects such as “digestive system symptoms and bleeding,” and also warns that the drug “can cause serious and fatal bleeding.”
Legal problems
According to Drug Watch, Pradaxa is manufactured by German pharmaceutical company Boehringer Ingelheim (BI) and was the company’s fourth largest grossing product in 2019 at $1.81 billion.
In November of 2020, Reuters Legal reported that BI had completed a global settlement resolving 2,935 lawsuits against the company “alleging that the drugmaker failed to warn patients about the bleeding risks of its blood thinner Pradaxa.”
The terms and amount of the global settlement were not publicly disclosed, but the article noted that, “In 2014, Boehringer agreed to pay about $650 million to resolve about 4,000 Pradaxa product liability lawsuits largely pending in federal court.”
Vaccines for kids
On Oct. 29, the FDA approved an Emergency Use Authorization to distribute the pediatric version of the Pfizer-BioNTech Messenger RNA vaccine to children aged 5 to 11 after the Administration’s 18-person Vaccines and Related Biological Products Advisory Committee voted 17 in favor and 1 abstention to approve the application.
During the meeting, conducted on Zoom, panelist Eric Rubin told listeners, “We’re never gonna learn about how safe the vaccine is until we start giving it…That’s just the way it goes. That’s how we found out about rare complications of other vaccines…”
In Canada, on Oct. 21, federal agency Public Services and Procurement Canada (PSPC) announced it had purchased 2.9 million doses of Pfizer’s pediatric injection to dose the 2,879,112 children aged 5 to 11 residing in the country in anticipation of a rubber stamping of Pfizer’s application.
Vaccinating children, however, is a popular prospect with Canadians. An Oct. 18 study by the Angus Reid Institute found that only 23 percent of respondents said they would spare their children from novel COVID vaccination.
Desaturation and clumping
A simple experiment conducted and broadcast by American scientist Dr. Richard Fleming and Japan-based scientist Kevin McCairn on Oct. 25 showed the effect of the Pfizer mRNA COVID vaccine when applied to human blood under the microscope.
Fleming and McCairn compared what McCairn’s blood looked like under the microscope by itself, what happened when normal saline solution was added, and what happened when the Pfizer vaccine was added.
The duo found that after the vaccine was dropped onto a slide of the scientist’s blood, it immediately began to discolor, indicating oxygen desaturation had occurred. Additionally, they recorded immediate and visible blood clotting.
In examining what the Pfizer vaccine looked like under the microscope, the pair also found the dose was littered with a variety of crystalline and fibrous structures, which Fleming described as “garbage” arising from a shoddy manufacturing process. The pair compared what the Pfizer injection looked like under the scope compared to how normal saline appeared, and the contrast was striking.