EDITOR'S PICK
-
‘China has been poisoned for over a century’: Statements From the Tuidang Movement (April 2024)
-
‘The CCP is the greatest enemy of the Chinese nation’: Statements From the Tuidang Movement (March 2024)

Why Millions of People Celebrate May 13 as World Falun Dafa Day
Latest
- Democracy’s Double-Edged Sword: How Ancient Greece Embraced the Lost Tool of ‘Ostracism’
- Japan’s Nippon Steel Committed to Closing US Steel Plant Deal by Year-end
- Hong Kong Court Formally Bans Protest Anthem ‘Glory to Hong Kong’
- Russia Detains Two US Citizens, Including American Soldier
- India’s Modi Aims for Supermajority in Upcoming General Election
FEATURED
-
EU Leaders Meet Xi Jinping in Paris, Express Concern Over Beijing’s Trade Practices
-
How the CCP Inflates China’s GDP
-
Pro-CCP Sogavare Steps Down as Leader of the Solomons, Dealing Major Blow to Beijing
-
‘The CCP Does Not Represent China’: Falun Gong Practitioners Commemorate 25th Anniversary of Appeal in Beijing
-
US Considering Sanctions on Chinese Banks for Russia Dealings, But No Concrete Plans Yet
-
Biden Proposes Tripling Tariffs on Chinese Steel, Aluminum to Counter Beijing’s Product Dumping

Destruction of Ancestral Temple in Southern China Brings 4,000 People Out in Protest
Lifestyle
-
Mind Over Matter: Train Your Brain and Transform Your Life
-
New York’s Hidden Treasure – Beautiful, Ethical and Economical Herkimer Diamonds
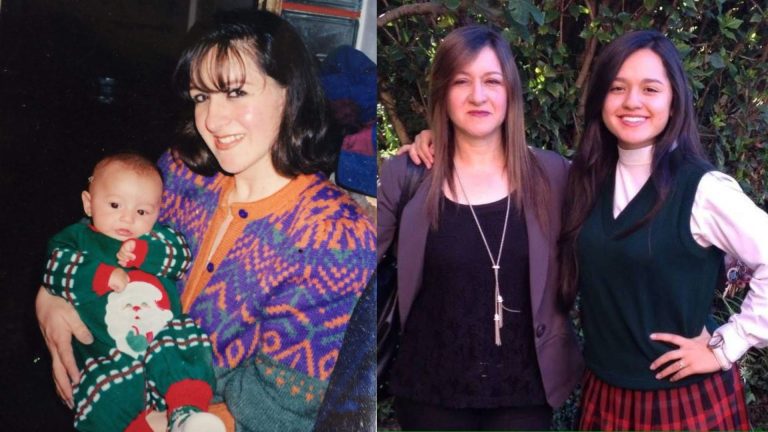
After Eight Mother’s Days Without Her, I Appreciate My Mom More Than Ever
Being a long-distance daughter has taught me valuable lessons about the essence of motherhood.
POLICY & POLITICS

FLUSHING, New York — On May 8, Kenneth Chiu, a candidate for the 25th New York State Assembly District, accompanied by his legal team, brought forth allegations of voter fraud involving one of his opponents, Steven Wang, at a press conference. Held at Bodhi Village in Queens, Chiu and his
-
Jose Perez: A Culinary Visionary Transforming American Dining
Jose Perez, an unassuming entrepreneur with a passion for flavor, has quietly
-
TikTok Sues to Overturn US Law Forcing Divestment From ByteDance
In the latest chapter of ongoing tensions between TikTok and the U.S.
-
NY: Dozens of Jewish Sites Receive Disturbing Bomb Threat Text
On May 4, more than two dozen synagogues and other Jewish institutions
- How NYC’s Business Improvement Districts Are Helping Small Businesses Thrive
- NY: This Broadway Hotel Has Been Quietly Operating as a Migrant Shelter for Over a Year
- Thousands Arrested as Student Protests Spread, Grow Violent
- Meet Kenneth Paek: Veteran, Policeman, and Advocate Vying for NY’s 25th Assembly
- Oil Prices Set for Steepest Weekly Drop in Three Months
- In China, Blinken Lays Out America’s Red Lines on Russia, Other Tensions
- Arizona Rancher George Alan Kelly Will Not Be Retried for Death of Mexican Migrant
- American Taxpayers Paying $451 Billion Annually to Address Southern Border Crisis
- ‘It moved my spirit’: See Why Theatergoers Are Enchanted With Shen Yun

On Friday, May 3, a new incursion of Communist Chinese military aircraft was detected by the Taiwan Defense Ministry in the Taiwan Strait area, with airplanes crossing the median line between the island and continential Asia. Beijing reported that its People’s Liberation Army (PLA) Navy carried out combat exercises with
-
Highway Collapse in Southern China Kills 48 as Poor Weather Hampers Rescue Efforts
In the early hours of May 1, disaster struck southeastern China when a section of a four-lane highway
-
Tibetans Who Sold Their Homes to Chinese Buyers Say Compensation Was Too Low
After selling their land to Chinese businessmen, Tibetan families are now reporting that they are not being compensated

Published with permission from LuxuryWeb Magazine In a landmark decision, Spain's Culture Ministry has announced the cancellation of the "Annual Bullfighting Award," sparking considerable controversy among conservative circles who cherish this centuries-old tradition as a quintessential Spanish cultural art form. “It is a new social and cultural reality in Spain,”
-
Russia Starts Drills With Tactical Nuclear Weapons in Response to ‘Provocative Statements’ From West
On Monday, May 7, the Russian military issued the deployment of tactical
-
Australia’s Defense Minister Denounces China Over Aerial Confrontation
Australia’s defense minister Richard Marles said in a statement on May 6
-
Flooding in Southern Brazil Claims at Least 39 Lives After Torrential Rains
Brazilian authorities of the country's southernmost Rio Grande do Sul state said
- Pandeli Locandasi: A Culinary Beacon Above Istanbul’s Vibrant Spice Bazaar
- Blinken in China: Calls for ‘Level Playing Field,’ Resolution Over Trade Disputes
- Moroccan Charm: A Timeless Journey Through Marrakech’s Ancient Medinas
- Taiwan to Remove Statues of Nationalist Chinese Leader Chiang Kai-shek
- Biden Signs Law Granting Massive $95 Billion Foreign Aid Package, Passes TikTok Bill
- Caribbean Treasure: Discovering the Rich Heritage of Ron Barceló Imperial Rum
- China’s Overseas Product Dumping: Addressing Unfair Competition Caused by Systemic Differences
- Charges Laid One Year After Historic Canadian Gold Heist
- A Journey Through Morocco: How to Master the Region’s Delicious Culinary Traditions

We have all experienced so called "gut feelings" – when we suddenly feel uneasy about something that rationally seems fine or, conversely, have firm confidence in something that has a high risk of failure. These puzzling flashes of insight are also referred to as intuition, hunches or sixth sense. They
-
Baoding Balls – They Do Much Better Than Bounce
 Baoding balls may have been invented in China’s city of the same name, located in Hebei Province, but the Chinese characters reveal another layer of meaning. Bao (保) carries the meaning “to protect, defend, or ensure,” while ding (定) usually means “to fix, or make definite.” In China, these specialized
Baoding balls may have been invented in China’s city of the same name, located in Hebei Province, but the Chinese characters reveal another layer of meaning. Bao (保) carries the meaning “to protect, defend, or ensure,” while ding (定) usually means “to fix, or make definite.” In China, these specialized
-
Innovation, Sustainability Shine at Natural Products Expo in California
ANAHEIM, California — The Natural Products Expo West stands out as a premier trade show within the natural,
-
For Powerful Traditional Remedies, Know and Grow Medicinal Herbs (G) Geranium
Geraniums are among the long list of baffling botanical nomenclature. While the beautiful showy flowers that are purchased

Do you suffer chronic symptoms that can’t be pinned down to any specific cause? Gastrointestinal disorders, headaches, hives, congestion, and itchy eyes can all be caused by histamine intolerance. Like gluten intolerance a couple decades ago, this condition is just gaining recognition and becoming a topic of study. Because it
-
For Powerful Traditional Remedies, Know and Grow Medicinal Herbs (F) Feverfew
Sprightly and tenacious, feverfew is a member of the world’s largest and most diverse plant family — Asteraceae.
-
The Power of Optimism – The Key to Longevity and Health
Life is hard, and staying positive is even harder. There are those who, despite sporting the scars of

Recent research has surfaced suggesting a major change in the themes of Mandarin song lyrics — particularly those performed by and aimed at Chinese women. Where love and romance were once common, now there seems to be a call for freedom. According to a team led by Wenbo Wang, business
-
The Sweet Satisfaction of Making Peace With Dessert
While people have been enjoying sweets for thousands of years, the concept of dessert has gradually evolved from
-
‘Extraordinary artistry’: Shen Yun Delights Theatergoers in Sold-Out Lincoln Center Shows
NEW YORK, New York — After kicking off a highly anticipated 14-show run at the prestigious David H.



